BALVERSA® is an oral kinase inhibitor targeting FGFR1
BALVERSA® is a kinase inhibitor that binds to and inhibits enzymatic activity of FGFR1, FGFR2, FGFR3, and FGFR4 based on in vitro data.
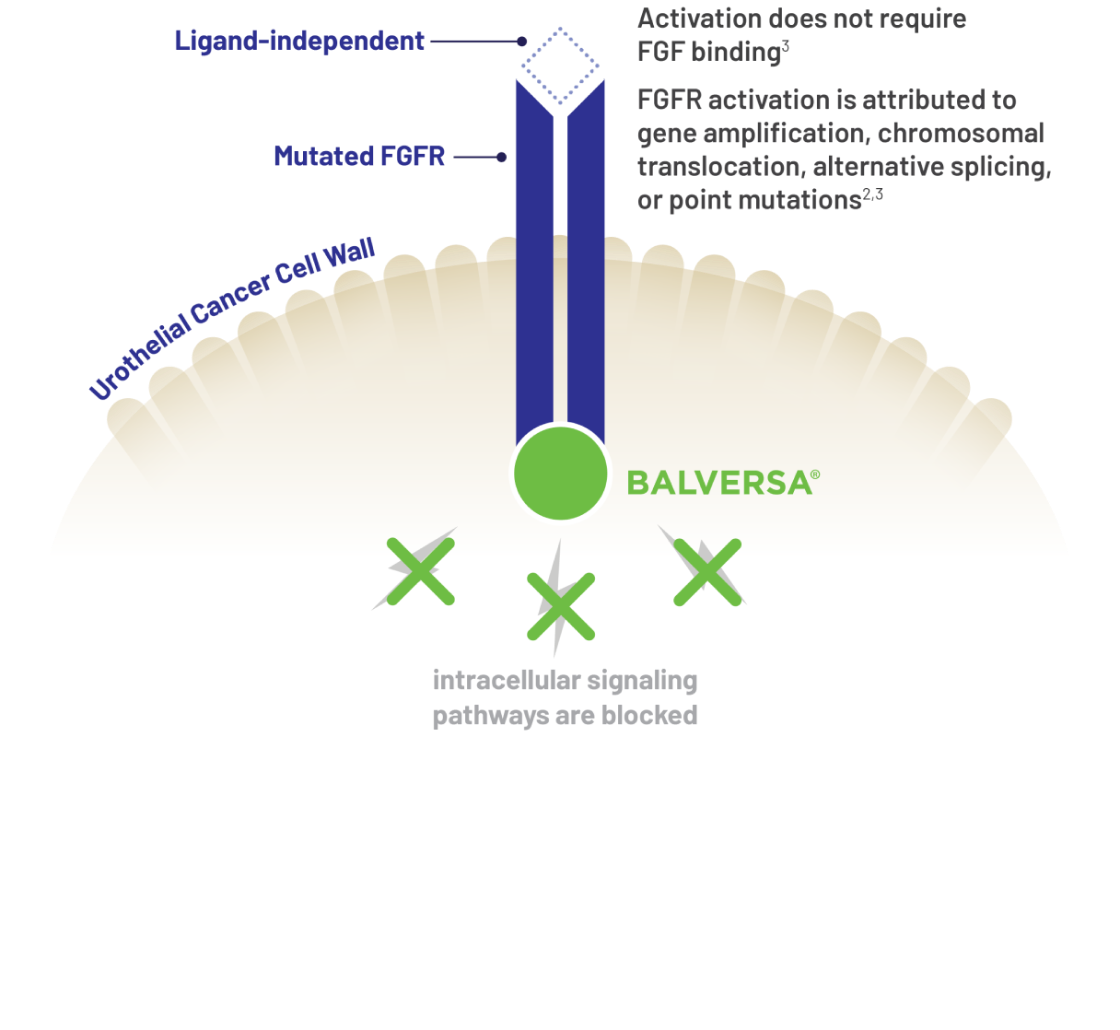
Because of the role of FGFR gene alterations in the deregulated proliferation of certain tumors, BALVERSA®, a small molecule inhibitor of FGFR, was developed.2 Molecular testing is required for patients to be prescribed BALVERSA®, because it is a targeted therapy that interacts with FGFR gene alterations. BALVERSA® is the first FDA-approved targeted kinase inhibitor of FGFR3 genetic alterations for the treatment of metastatic urothelial carcinoma.1*
BALVERSA® inhibited FGFR phosphorylation and signaling and decreased cell viability in cell lines expressing FGFR genetic alterations, including point mutations, amplifications, and fusions.1
BALVERSA® demonstrated antitumor activity in FGFR-expressing cell lines and xenograft models derived from tumor types, including bladder cancer.1
BALVERSA® binds to and inhibits enzymatic activity of FGFR1, FGFR2, FGFR3, and FGFR4 based on in vitro data1*
Test for FGFR alterations/mutations to identify appropriate patients
The FDA-approved companion diagnostic test detects clinically actionable FGFR genetic alterations from formalin-fixed paraffin-embedded urothelial tumor tissue.4,5
The approved companion diagnostic is available in various national and regional molecular pathology testing labs.
Find a testing lab located near you.
*See Mechanism of Action (section 12.1) in the full BALVERSA® Prescribing Information.
FDA = U.S. Food and Drug Administration; FGF = fibroblast growth factor; FGFR = fibroblast growth factor receptor.
References
1. BALVERSA® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 2. Perera TPS, Jovcheva E, Mevellec L, et al. Discovery and pharmacological characterization of JNJ-42756493 (erdafitinib), a functionally selective small-molecule FGFR family inhibitor. Mol Cancer Ther. 2017;16(6):1010-1020. 3. Karkera JD, Cardona GM, Bell K, et al. Oncogenic characterization and pharmacologic sensitivity of activating fibroblast growth factor receptor (FGFR) genetic alterations to the selective FGFR inhibitor erdafitinib. Mol Cancer Ther. 2017;16(8):1717-1726. 4. U.S. Food and Drug Administration. List of cleared or approved companion diagnostic devices (in vitro and imaging tools). Accessed January 17, 2023.https://www.fda.gov/medical-devices/vitro-diagnostics/list-cleared-or-approved-companion-diagnostic-devices-vitro-and-imaging-tools 5. therascreen® FGFR RGQ RT-PCR Kit, Instructions for Use (Handbook). HB-2561-001, version 1, April 2019. Germantown, MD: QIAGEN N.V.